27 April 2016
4QFY16 Results Update | Sector: Healthcare
Biocon
Sell
BSE SENSEX
26,064
Bloomberg
Equity Shares (m)
M.Cap.(INR b)/(USD b)
52-Week Range (INR)
1, 6, 12 Rel. Per (%)
Avg Val, (INR m)
Free float (%)
S&P CNX
7,980
BIOS IN
200.0
114.2/ 1.7
581/397
20/29/35
313
39.3
CMP: INR571
TP:INR490 (-14%)
Mixed results; Development on Biosimilars portfolio is key
Financials & Valuation (INR b)
Y/E Mar
2016 2017E 2018E
Sales
34.5
39.4
46.5
EBITDA
7.9
9.4
11.4
Net Profit
4.6
5.2
6.1
Adj. EPS (INR)
23.2
26.2
30.6
EPS Gr. (%)
15.5
13.0
16.5
BV/Sh. (INR)
195.2 213.8 235.4
RoE (%)
11.9
12.3
13.0
RoCE (%)
21.1
12.6
14.0
P/E (x)
24.6
21.8
18.7
P/BV (x)
2.9
2.7
2.4
Estimate change
TP change
Rating change
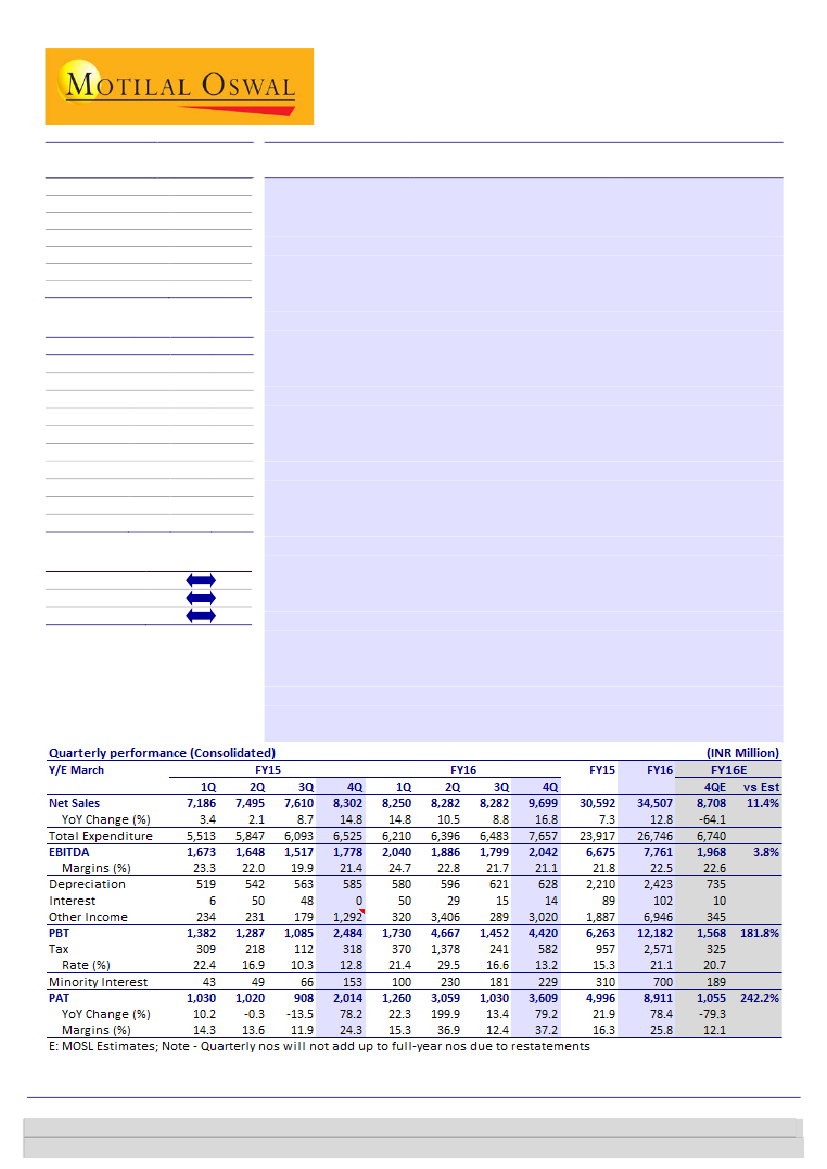
Biocon’s 4QFY16 PAT at INR3.6b was boosted by a one-time deferred income of
INR2.6b, excluding which the PAT came in line with our estimate. Topline grew 17%
YoY (11% beat) to INR9.7b, aided by a strong growth in Syngene. EBITDA stood at
INR2b, while EBITDA margin was lower at 21.1% due to higher R&D expenses.
CRO leads in terms of Revenue:
The CRO/Syngene business continued to
witness strong momentum with a 32% YoY revenue growth, while the
Biopharma business (excluding India) also reported a higher than expected
growth (up 8% YoY) due to growing sales of biosimilars in emerging markets.
The India branded business remained sluggish with a growth of 3.4% YoY,
affected by portfolio rationalization and weak sales of in-licensed products.
Overall, we project a 15% YoY revenue growth over FY15-18E, supported by a
20% YoY growth in the Syngene business.
R&D investment at record high:
The EBITDA margin at 21.1% was down 36bp
YoY, owing to a spike in R&D expenses (10.3% of sales as against 6.7% in
4QFY15). We expect further margin improvement to be restricted by increased
R&D activities on account of higher number of ANDA filings and progression of
biosimilar projects in clinical trials (five molecules in Phase 3 clinical trial). We
project an EBITDA CAGR of 20% on the back of a slightly improved EBIDTA
margin at 24-25% by FY18E.
Biosimilars sales in regulated markets to kick off only post FY19:
Though Glargine
approval for Japan market is encouraging, approvals for US and EU region would be
key growth driver for the company and launches in these regulated market is only
possible post FY19. Until then, the company’s profitability is likely to remain under
pressure due to high R&D expenses, increasing capex and lower utilization levels of
new capacities. We maintain our SELL rating on the stock with a target price of
INR490 as the earnings outlook for Biocon remain modest over FY17/18E (15% EPS
CAGR as against ~20% for its peers), and big investment projects like the
biosimilars/Malaysia insulin plant are unlikely to generate significant revenue
before FY18.
Kumar Saurabh
(Kumar.Saurabh@MotilalOswal.com); +91 22 3982 5584
Amey Chalke
(amey.chalke@motilaloswal.com); +91 22 39825423
Investors are advised to refer through important disclosures made at the last page of the Research Report.
Motilal Oswal research is available on www.motilaloswal.com/Institutional-Equities, Bloomberg, Thomson Reuters, Factset and S&P Capital.